|
The oxidation number of a monatomic ion is the charge on that ion. 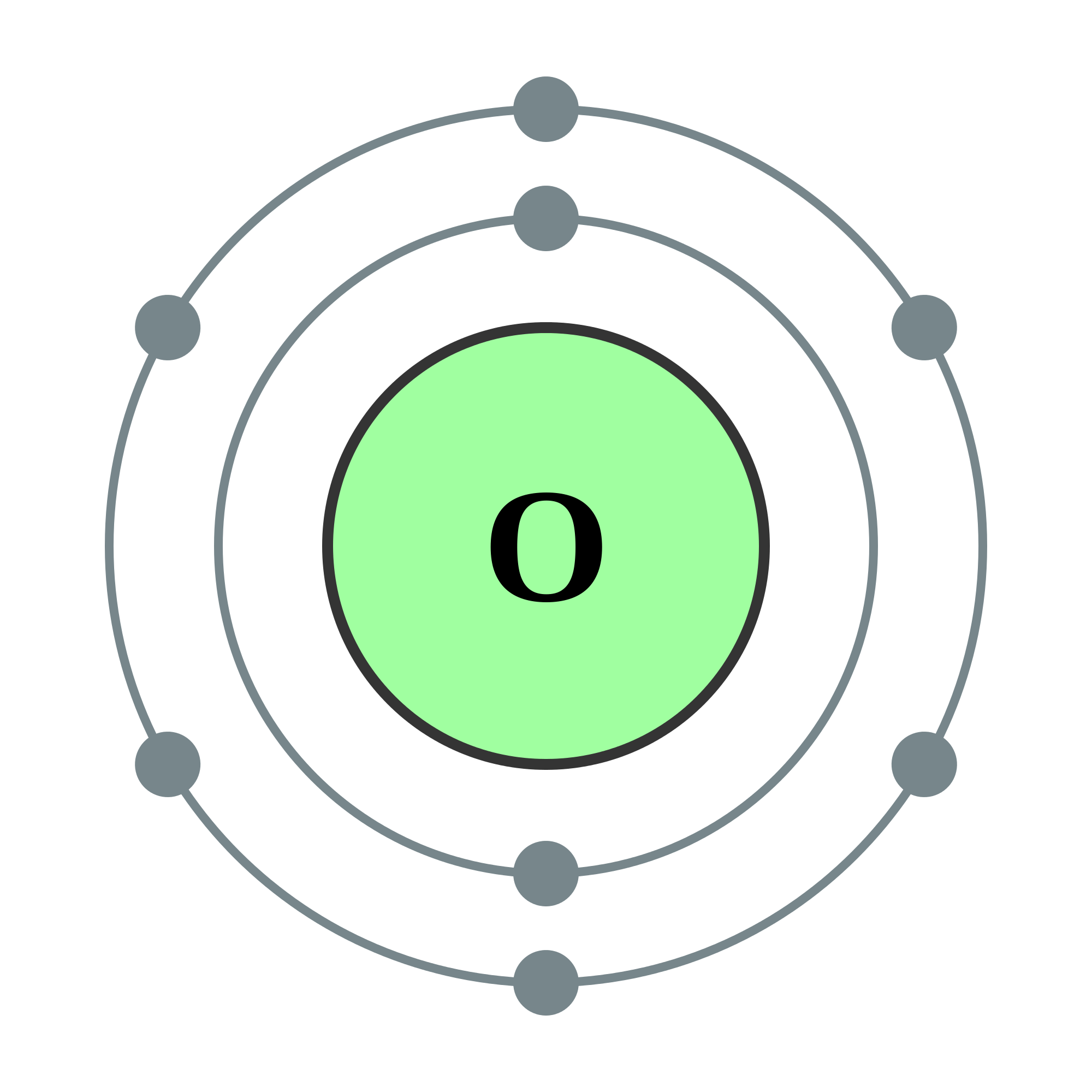
Since oxygen has an atomic number of \ (8\), a neutrally charged oxygen atom has \ (8\) protons. The oxidation number of an uncombined element is 0. Notice only the number of oxygen atoms changed, the charge did not. 
It is the 8th periodic element with a molar mass of \ (16.00\) grams, and it typically has six valence electrons. charge I combines with three oxygens and has a 1- charge. In the SI system of units, the value of the elementary charge is exactly defined as e īy combining the best measured value of the antiproton charge (below) with the low limit placed on antihydrogen's net charge by the ALPHA Collaboration at CERN. Oxygen is one of the most common and useful elements in the world, particularly because a good portion of what humans breathe is oxygen. 
The elementary charge, usually denoted by e, is a fundamental physical constant, defined as the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. F gains 1 e- and O gains 2 e-s, and O gain e-s to attain the configuration of Ne thus, the halogens tend to -1 charges and the oxygen family tends to a -2. Living on Mars requires the ability to synthesize chemicals that are essential for survival, such as oxygen, from local Martian resources.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
